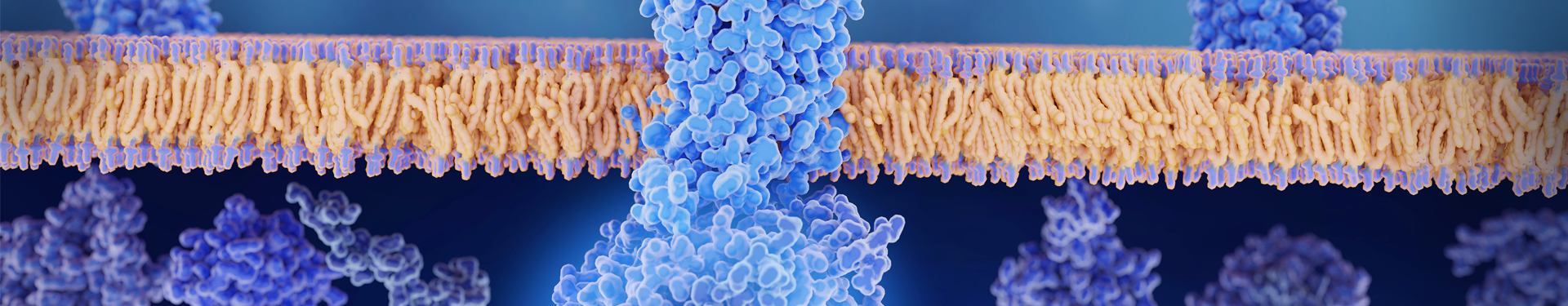
Therapeutic Use of IgG protease
IgG Protease as Pre-transplant Treatment
Imlifidase (Idefrix) is a recombinant immunoglobulin G (IgG)-degrading enzyme derived from Streptococcus pyogenes, which is also known as IdeS. The active ingredient of Imlifidase (IdeS) is produced in Escherichia coli (E. coli) cells using recombinant DNA technology. IdeS cleaves IgG to form F(ab')2 and Fc fragment, thereby eliminating Fc-dependent effector functions such as antibody-dependent cell-mediated cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC). Imlifidase is developed by Hansa Biopharma AB. EMA approved Imlifidase as an immunoglobulin inhibitor as a treatment strategy for kidney transplant recipients with positive crossmatch.
IgG Protease for Overcoming AAV Immunity
Adeno-associated virus (AAV) is a promising platform for target gene delivery. It has been previously shown that even a small amount of neutralizing antibodies (NAbs) against AAV can thoroughly eliminate transduction of high-titer AAV vectors. Furthermore, NAb is already present in a significant proportion of AAV-exposed individuals. Thus, the immunogenicity severely limits the potential application of AAV-mediated gene therapy.
Thus, gene therapy has been the focus of IgG proteases for many years. For instance, IdeZ is a type of IgG derived from Streptococcus equi subspecies zooepidemicus. For passively immunized animals in the presence of Nabs to AAV, prophylactic administration of it can prevent antibody neutralization as well as rescue AAV transduction. Another IgG protease from Streptococcus pyogenes, known as IdeS, also enables efficient transduction in nonhuman primates with pre-existing NAbs, and even AAV readministration.
In 2020, Hanza awarded Sarepta an exclusive license to evaluate imlifidase as pre- administration to enable Sarepta’s gene therapy treatment based on AAV vector. They will assess if imlifidase pre-treatment could overcome AAV immunity in patients with Duchenne muscular dystrophy (DMD) and limb-girdle muscular dystrophy (LGMD) with pre-existing antibodies to AAV.
IgG Protease as Reagent
IdeS is a specific IgG degrading enzyme, which can recognize and cleave IgG at the hinge region, producing F(ab')2 or Fab fragments and the Fc fragment. IdeS, or IgG protease, is widely used in monoclonal antibody (mAb) subunit analysis.
Yaohai Bio-Pharma Offers One-Stop CDMO Solution for IgG protease
IgG protease Pipeline
|
Generic Name
|
Brand Name/ Altermative Name
|
Expression System
|
Indications
|
Manufacturer
|
R&D Stage
|
|
Idefirix
|
imlifidase
|
E. coli
|
Transplant Rejection
|
Hansa Biopharma AB
|
Approval
|
|
KJ103
|
Pending Update
|
Pending Update
|
Kidney transplant rejection
|
Shanghai Bao Pharmaceuticals
|
Phase I
|
|
JZB-34
|
AAFP
|
Pending Update
|
Allergic asthma
|
Shanghai Kexin Biotech
|
Phase I
|
Reference:
[1] Al-Salama ZT. Imlifidase: First Approval. Drugs. 2020 Nov;80(17):1859-1864. doi: 10.1007/s40265-020-01418-5.
[2] Li X, Wei X, Lin J, Ou L. A versatile toolkit for overcoming AAV immunity. Front Immunol. 2022 Sep 2;13:991832. doi: 10.3389/fimmu.2022.991832.

 EN
EN
 AR
AR
 HR
HR
 CS
CS
 DA
DA
 NL
NL
 FI
FI
 FR
FR
 DE
DE
 EL
EL
 IT
IT
 JA
JA
 KO
KO
 NO
NO
 PL
PL
 PT
PT
 RO
RO
 RU
RU
 ES
ES
 SV
SV
 IW
IW
 ID
ID
 LV
LV
 LT
LT
 SR
SR
 SK
SK
 SL
SL
 UK
UK
 VI
VI
 ET
ET
 HU
HU
 TH
TH
 TR
TR
 FA
FA
 AF
AF
 MS
MS
 BE
BE
 MK
MK
 UR
UR
 BN
BN