Article

Flexible CDMO Solutions Leading Innovation Through Agility
Mar 26, 2025Biologics have revolutionized modern medicine with targeted therapies that address complex diseases and unmet medical needs. However, their development is fraught with challenges—from unstable molecular structures and the reliance on living sys...
Read More-

Microbial CDMO Drives Revolutionary Wave of Biologics Manufacturing
Mar 25, 2025The biopharmaceutical industry is undergoing a transformative period known as the “fourth revolutionary wave” of biologics manufacturing. This new era centers on automation, process optimization, and flexible production approaches. Tradit...
Read More -

Global CDMO Landscape: Opportunities and Challenges
Mar 18, 2025Contract Development and Manufacturing Organizations (CDMOs) offer comprehensive drug development and manufacturing services, catalyzing the journey from preclinical research to commercialization. By partnering with CDMOs, biotech firms can leverage ...
Read More -
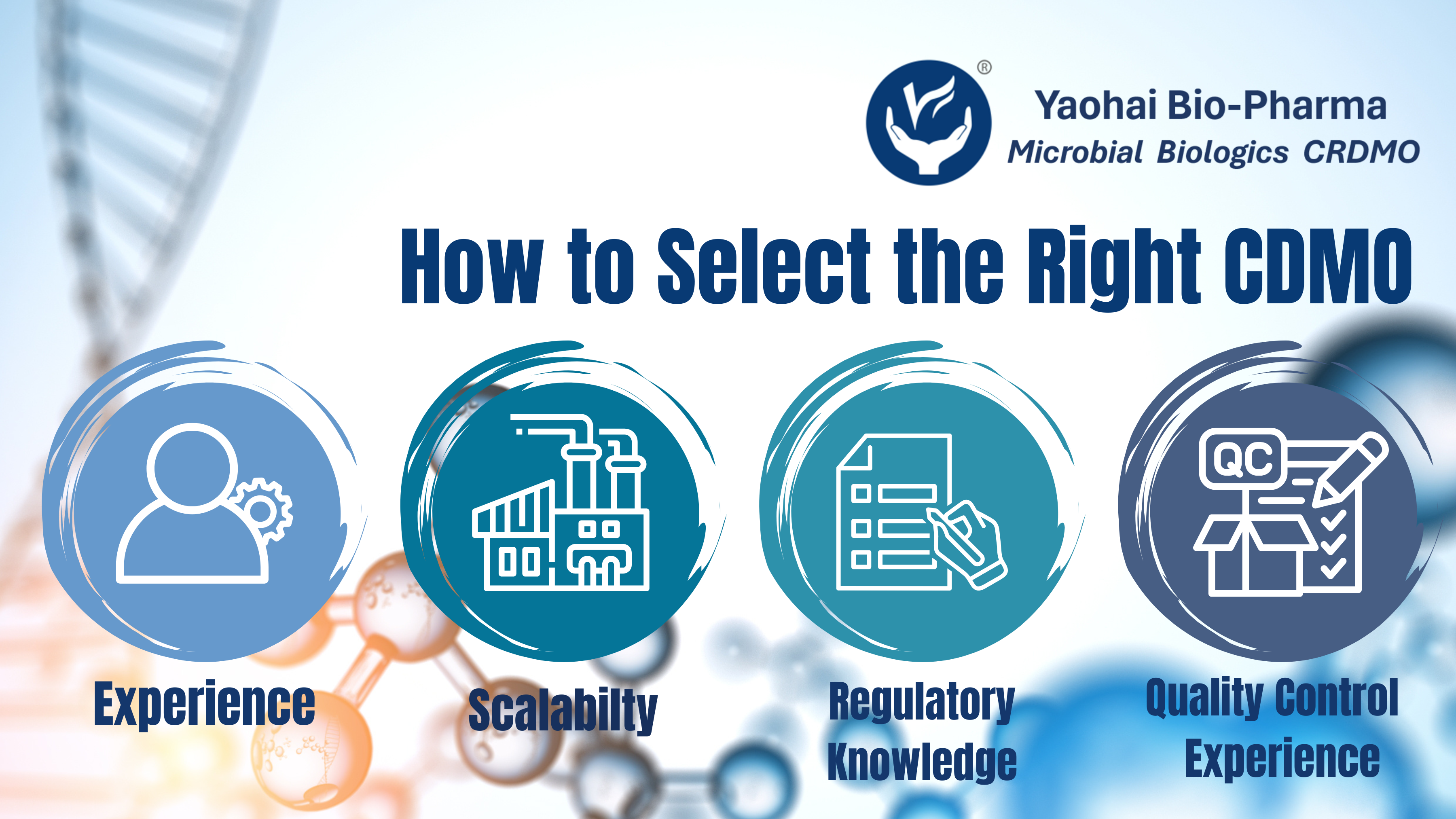
Key Considerations When Choosing the Right CDMO Partner
Mar 13, 2025In the rapidly evolving landscape of pharmaceutical development, selecting the right Contract Development and Manufacturing Organization (CDMO) partner is crucial to the success of your drug product. With over a decade of industry experience, Yaohai ...
Read More -

Navigating the Complex Regulatory Environment with a CDMO
Mar 11, 2025The biopharmaceutical industry faces unprecedented challenges navigating evolving regulatory frameworks, particularly as global standards tighten and regional requirements diversify. Partnering with a Contract Development and Manufacturing Organizati...
Read More -

Microbial CDMO is Becoming the Go-To Solution for Biotech
Mar 06, 2025Microbial Contract Development and Manufacturing Organizations (CDMOs) specialize in harnessing microbial fermentation systems to produce a wide range of biological products, such as proteins, peptides, and vaccines. Unlike mammalian CDMOs, which rel...
Read More -
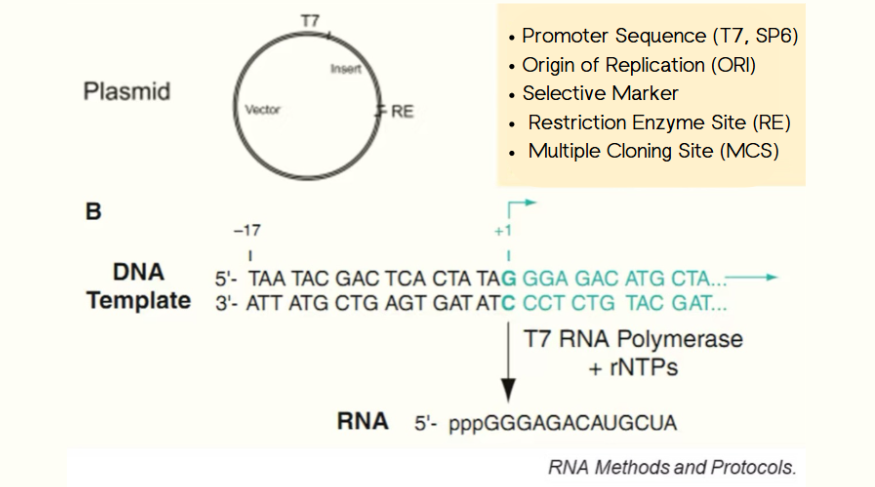
mRNA Plasmid Templates: Key Elements and Design Strategies
Mar 05, 2025Creating mRNA for vaccines or therapies starts with a well-designed plasmid template. Here’s a quick guide to the key elements and how they’re optimized for success. The Promoter: Where Transcription Begins The T7 promoter is the most pop...
Read More -

Exploring the expression systems in CDMO
Mar 04, 2025Contract Development and Manufacturing Organization (CDMO) plays a pivotal role in the biopharmaceutical industry by providing specialized services for biologics development. One critical factor in this process is selecting the right expression syste...
Read More -

Help You Quickily Understand CMO, CDMO, and CMO
Feb 27, 2025In the pharmaceutical and biotechnology industries, selecting the appropriate partner for drug development and manufacturing is crucial. While Contract Research Organization (CRO) and Contract Manufacturing Organizations (CMO) provide valuable servic...
Read More -

What is a CDMO? Understanding the Role and Benefits
Feb 26, 2025A Contract, Development, and Manufacturing Organization (CDMO) is a company that offers comprehensive services to the pharmaceutical and biotechnology industries, including microbial strain engineering, microbial cell banking, process development, pi...
Read More -
E. coli: A Versatile Engine for Drug Innovation
Feb 25, 2025Since the advent of recombinant DNA technology, Escherichia coli (E. coli) has emerged as a cornerstone of biopharmaceutical production. Its rapid growth, simple nutrition, and well-understood genetics enable precise engineering for therape...
Read More -
VLP Vaccine: A Novel Approach to Combat Mosquito-Borne Viruses
Feb 19, 2025Virus-like particles (VLPs), non-infectious proteins mimicking viruses, are crucial in vaccine development. Texas Tech University researchers created a multivalent VLP vaccine targeting Chikungunya (CHIKV), Japanese Encephalitis (JEV), Yellow Fever (...
Read More -

Quality Control and Applications of Recombinant Proteins
Feb 18, 2025Quality control of recombinant proteins is crucial for the reliability and reproducibility of experimental data. Every step, from project design to production process, requires stringent quality control strategies. Quality Control Strategies The indu...
Read More -

Plasmid: A Small Circular DNA Molecule with Big Potential
Feb 14, 2025Plasmids, small circular DNA molecules, serve as essential vectors for DNA recombination in molecular biology research and gene therapy. The yield and quality of plasmids are crucial, with supercoiled proportion and endotoxin content being two signif...
Read More -

Optimizing the Purification Process for Recombinant Human Insulin
Feb 13, 2025In recent years, diabetes patient growth has driven insulin demand, but affordable insulin is in short supply. Efficient, economical insulin production is crucial. Mainly produced via Escherichia coli (E. coli) and yeast due to rapid growth and low c...
Read More -
Yeast cells for recombinant protein production
Feb 12, 2025Traditional methods for acquiring pharmaceutical raw materials, such as plant extraction or relying on natural organisms, face challenges like long growth cycles and limited yields. Microbial recombinant protein synthesis technology, particularly usi...
Read More -
Optimizing E. coli Cellular Integrity for Biopharma Production
Feb 06, 2025In the field of biopharmaceuticals, Escherichia coli (E. coli) plays a vital role as a host for the expression of recombinant proteins. The cellular integrity of E. coli is crucial for protein yield, quality, and production costs. This integrity prim...
Read More -
Codon Optimization Strategies
Feb 05, 2025There are 64 genetic codons, of which 60 encode 20 amino acids. Every organism used for protein expression or production (including Escherichia coli, yeast, mammalian cells, Pichia, plant cells, and insect cells) exhibits some degree of difference or...
Read More -
Overcoming Challenges in Therapeutic Protein Purification
Jan 23, 2025In recent decades, recombinant proteins have proven crucial in combating various diseases, demonstrating great potential. With advancements in genetic engineering, significant breakthroughs have been made in protein drug research. Technologies like f...
Read More -
IVT mRNA Preparation
Jan 22, 2025In vitro transcription (IVT) is the preferred method for preparing mRNA, capable of yielding micrograms to milligrams of mRNA on a laboratory scale. For research purposes, reagent suppliers have developed versatile IVT reaction systems suitable for m...
Read More
Hot News
-
Yaohai Bio-Pharma Passed EU QP Audit and Attains ISO Triple Certification
2024-05-08
-
BiotechGate, Online
2024-05-13
-
2024 WORLD VACCINE CONGRESS Washington
2024-04-01
-
CPHI North America 2024
2024-05-07
-
BIO International Convention 2024
2024-06-03
-
FCE COSMETIQUE
2024-06-04
-
CPHI Milan 2024
2024-10-08

 EN
EN
 AR
AR
 HR
HR
 CS
CS
 DA
DA
 NL
NL
 FI
FI
 FR
FR
 DE
DE
 EL
EL
 IT
IT
 JA
JA
 KO
KO
 NO
NO
 PL
PL
 PT
PT
 RO
RO
 RU
RU
 ES
ES
 SV
SV
 IW
IW
 ID
ID
 LV
LV
 LT
LT
 SR
SR
 SK
SK
 SL
SL
 UK
UK
 VI
VI
 ET
ET
 HU
HU
 TH
TH
 TR
TR
 FA
FA
 AF
AF
 MS
MS
 BE
BE
 MK
MK
 UR
UR
 BN
BN

