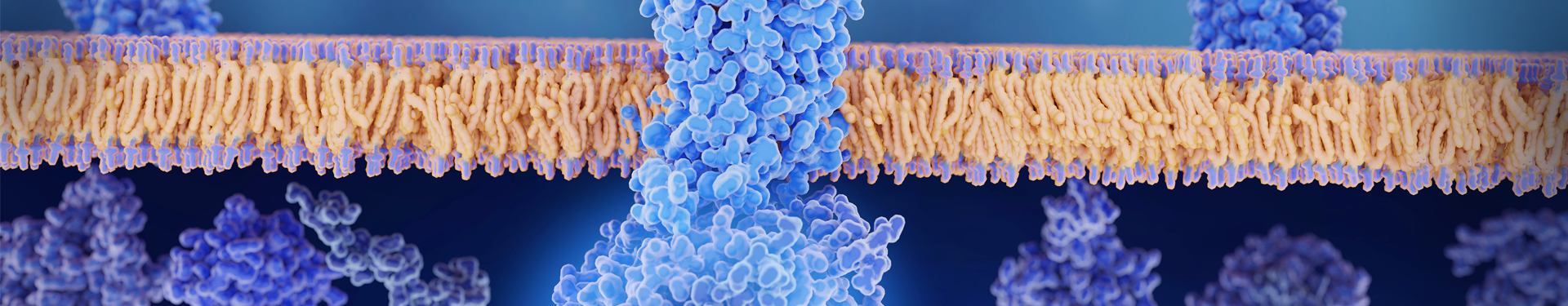
Circular RNAs (circRNAs) were first discovered as non-coding RNAs in 1979; it was not until 2015 that transcribed circRNAs were found in Drosophila, which led to the study of in vitro circRNAs for therapeutic and preventive purposes.
circRNAs are self-expanding and therefore do not have a conventional structure such as a 5' cap or 3' polyA tail. This structure makes them more stable and resistant to exonucleases like RNase R.
Structure Elements of Circular RNA (circRNA)
The template DNA for circRNA synthesis typically includes internal ribosome entry site (IRES), open reading frame (ORF), and others that are critical for in vitro circulation. For example, we designed a plasmid as in vitro transcript (IVT) template, which consists of homology arms, 3’ intron, 3’ exon, IRES, ORF, 5’ exon, and 5’ intron, to prepare circRNA by self-splicing methods.
Internal Ribosome Entry Site (IRES) performs important roles in the translation of cirRNAs. Encephalomyocarditis virus (EMCV) IRES, coxsackievirus B3 (CVB3) IRES, human rhinovirus B3 (HRV-B3) IRES and other IRES have been commonly utilized for the translation of the vitro synthesis of cirRNAs
In vivo cyclization of circRNA linear precursors provides an important step in circRNA synthesis, usually through chemical, enzymatic, and ribosome-mediated self-replication pathways. Known as a milestone in the field, the chemical approach for circRNA production, originated in 1988, is no longer used today because of its high cost, low yield, high number of by-products, and the fact that it is only suitable for cyclizing RNAs up to 70 nucleotides in length.
Enzymatic methods for cyclizing RNA are based on bacteriophage T4 enzymes or ribozymes such as T4 DNA ligase (T4 Dnl 1), T4 RNA ligase 1 (T4 Rnl 1), and T4 RNA ligase 2 (T4 Rnl 2). To form circular RNA by T4 bacteriophage enzymes, nucleoside monophosphates must be located at the 5' end and conjugated to the OH group at the 3' end of the RNA. Because nucleoside triphosphates are added during the reaction, IVT RNA contains guanosine triphosphate (GTP) at the 5’ end.
Ribozymes are RNA sequences that promote self-splicing by converting linear RNA molecules into circRNA without the need for additional enzymes. The self-assembly process involves two consecutive transesterification reactions at specific sites to make sure that the desired cirRNA products are formed. The group I intron self-splicing method, also known as PIE (intron and exon encapsulation), which allows the production of cirRNAs longer than 5 kb, has been extensively studied and has proven useful.
Application of Circular RNA (circRNA)
CircRNA Vaccines
Like linear mRNAs, circRNAs can be converted into certain proteins in target cells and induce the robust humoral and cellular immunity. Recently, there are some research teams who successfully developed circRNA vaccines to prevent COVID-19. Their results have not only possessed the benefits of linear mRNA vaccines, but also boasted a better stability and longer duration of protein expression than linear mRNA. Therefore, circRNA vaccines can induce adequate immune responses even at low doses.
In addition, some researchers think that circRNA vaccines, as next-generation mRNAs, may become effective tools in the future for combating common viral/bacterial diseases and major emerging infectious diseases, as well as for the treatment of cancer and other diseases.
CircRNA in CAR / TCR-T Therapy
As a pioneer in the field of circular RNA, ORNA has been developing in situ chimeric antigen receptor (CAR) therapy for in situ therapy that utilizes with ORN-101, a LNP-encapsulated circular RNA, to modulate immune cells in patients. ORN-101 presents high expression of the CAR driven by an optimized IRES element. In animal models, ORN-101 was shown to induce tumor suppression and destruction, suggesting that ORN-101-based anticancer therapies may interfere with conventional CAR-T cell therapy.
In addition, Zhang et al. evaluated the viability and therapeutic efficacy of circRNAs in antigen-specific T-cell receptor (TCR)-T therapies. They designed a circRNA encoding pp65-TCR-T targeting the pp65 epitope of cytomegalovirus (CMV). In addition, pp65-TCR is demonstrated to be expressed on primary T cells for over 7 days. In addition, circRNA-pp65-TCR-T cells specifically and consistently killed pp65- and HLA-expressing tumor cells and significantly extended the survival time of mice.
It was also shown that cells transfected with pp65 circRNA exhibited better immune responses compared to linear mRNA.
Yaohai’s Bio-Pharma One-Stop CRDMO Solution for Long-Coding RNA

 EN
EN
 AR
AR
 HR
HR
 CS
CS
 DA
DA
 NL
NL
 FI
FI
 FR
FR
 DE
DE
 EL
EL
 IT
IT
 JA
JA
 KO
KO
 NO
NO
 PL
PL
 PT
PT
 RO
RO
 RU
RU
 ES
ES
 SV
SV
 IW
IW
 ID
ID
 LV
LV
 LT
LT
 SR
SR
 SK
SK
 SL
SL
 UK
UK
 VI
VI
 ET
ET
 HU
HU
 TH
TH
 TR
TR
 FA
FA
 AF
AF
 MS
MS
 BE
BE
 MK
MK
 UR
UR
 BN
BN