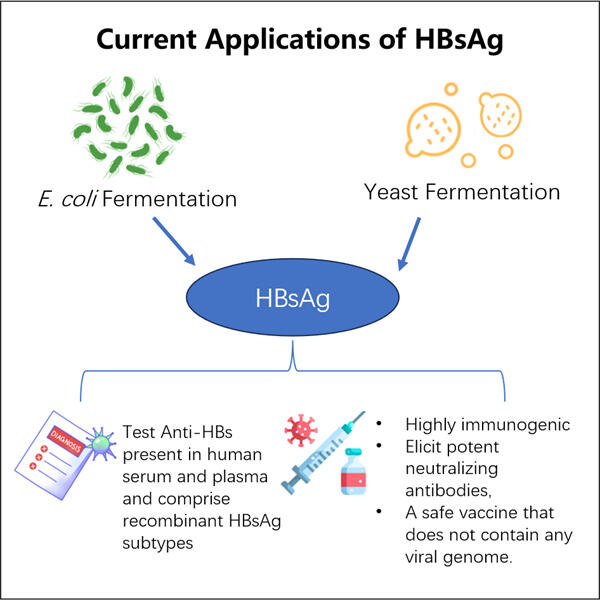
Yaohai je společnost, která se stará o lidi, kteří nejsou tak zdraví. Jsou to skupina lidí pracujících v pohotovosti 24 hodin denně na rozvoji nových léčeb pro běžné nemoci postihující lidi po celém světě. A jednou z hlavních zdravotních problémů, které bojují, je něco, co se nazývá HBV (běžně hepatitida B). Jedná se o jednoho z hlavních infekčních činitelů způsobujících vážné onemocnění a dlouhodobé důsledky. S jejich výzkumem si Yaohai myslí, že mohou pomoci změnit životy těm, kdo trpí touto nemocí.
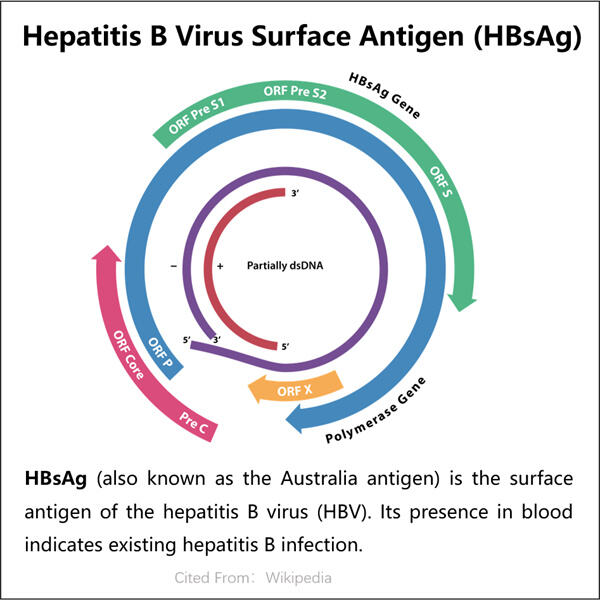
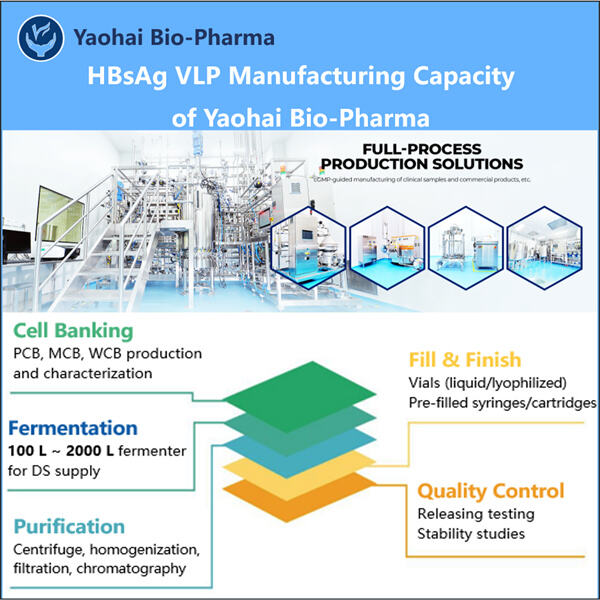
Jednou z klíčových snah Yaohai proti HBV je Výroba VLP bakteriofágu AP205 zvláštní vynález nazvaný HBsAg VLPs. Nepřítomnost držáku vzorku (k prevenci vzniku pěny navrchu): vzorky jsou brány přímo z RN (RNA genu; RNPs podle jiných uznávaných termínů). Tyto částice jsou podobné virům, ale nejsou to skutečné viry, takže vás nemohou vůbec nakazit. Yaohai doufá, že tyto VLPs naučí tělo rozpoznat a lépe odolávat viru HBV. Je to velmi vzrušující zpráva v boji proti HBV!

 EN
EN
 AR
AR
 HR
HR
 CS
CS
 DA
DA
 NL
NL
 FI
FI
 FR
FR
 DE
DE
 EL
EL
 IT
IT
 JA
JA
 KO
KO
 NO
NO
 PL
PL
 PT
PT
 RO
RO
 RU
RU
 ES
ES
 SV
SV
 IW
IW
 ID
ID
 LV
LV
 LT
LT
 SR
SR
 SK
SK
 SL
SL
 UK
UK
 VI
VI
 ET
ET
 HU
HU
 TH
TH
 TR
TR
 FA
FA
 AF
AF
 MS
MS
 BE
BE
 MK
MK
 UR
UR
 BN
BN