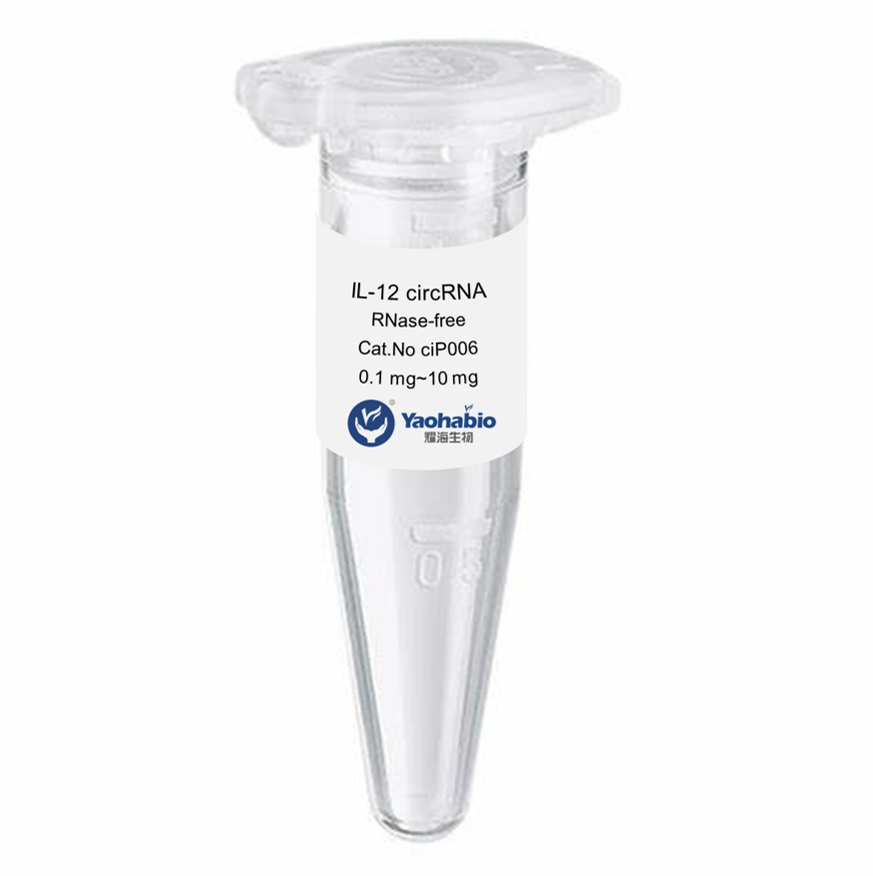
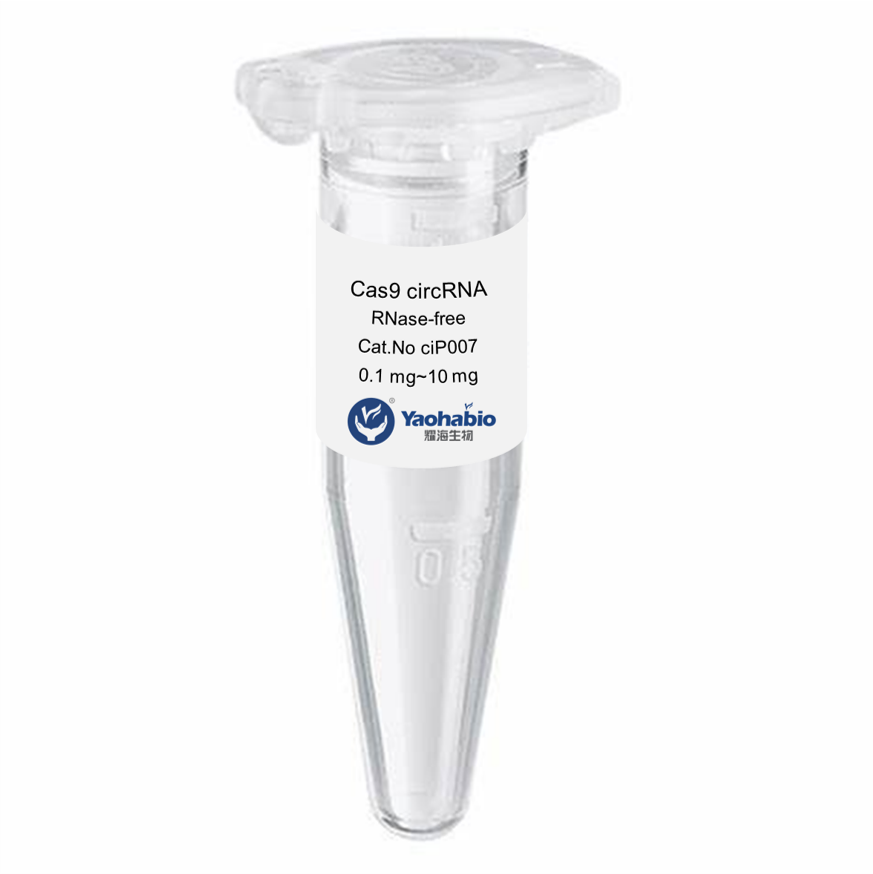
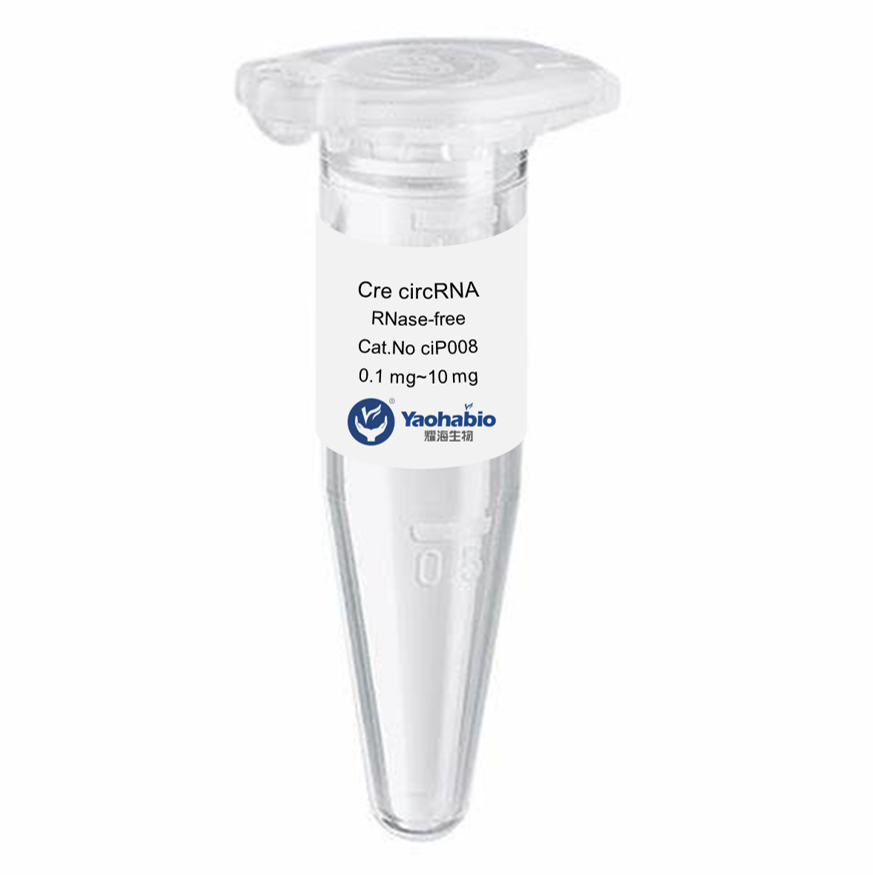
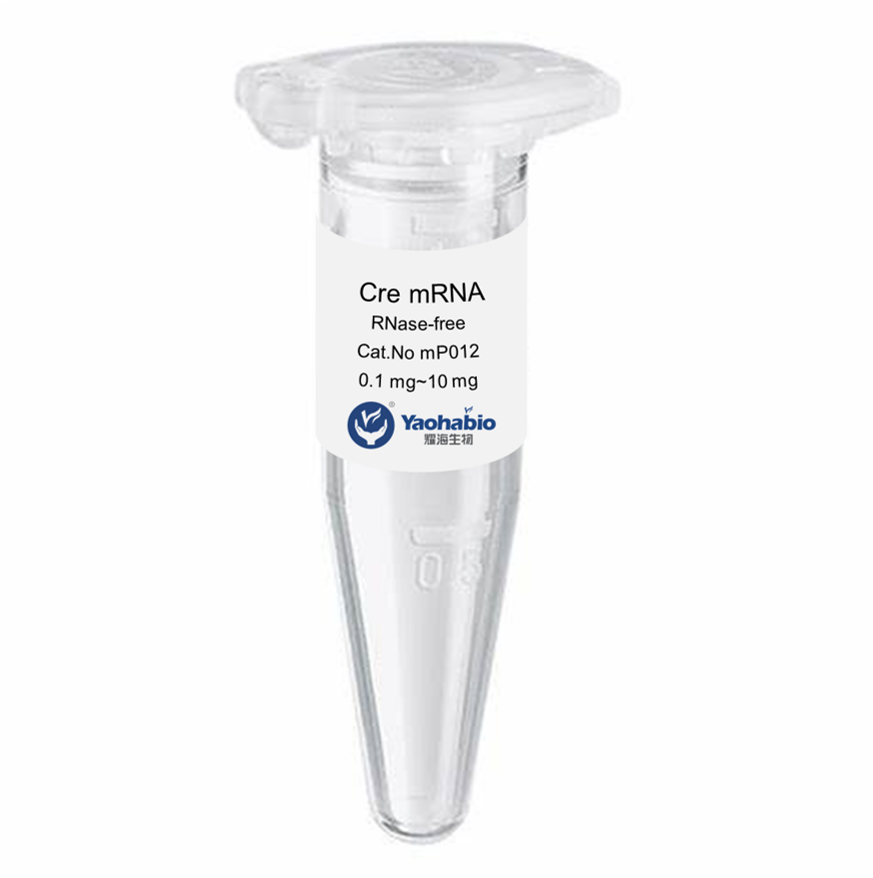
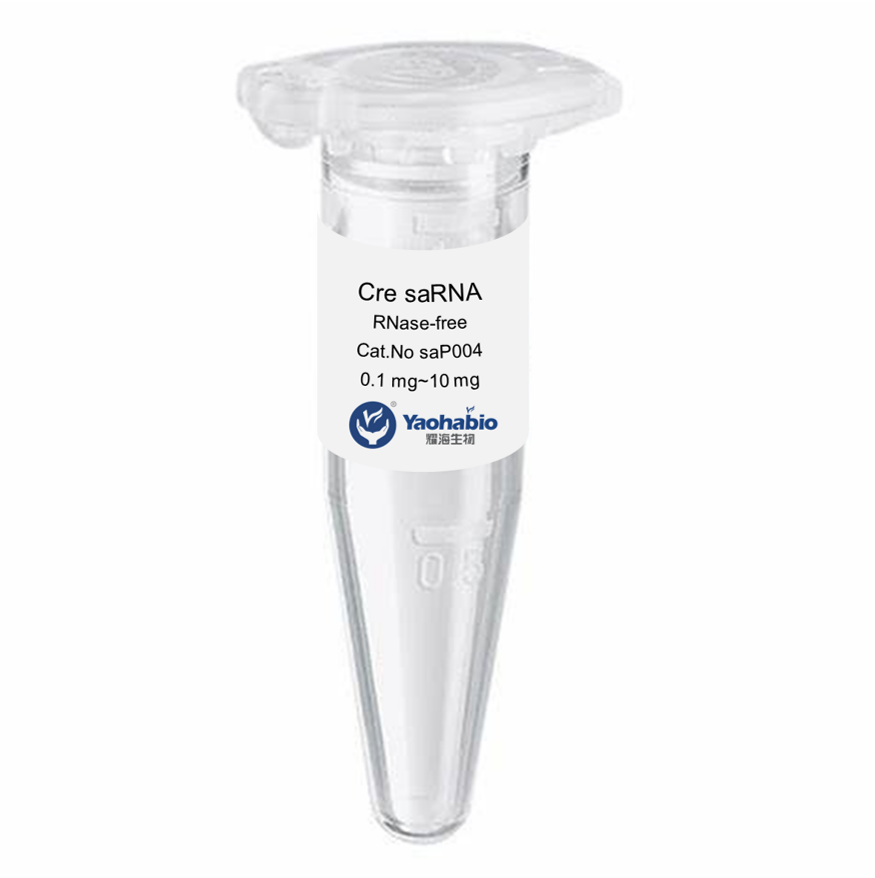
Customization, Efficiency & Cost-Effectiveness
Yaohai Bio-Pharma is experienced in biologicals derived from microbial sources. We provide customized RD solutions and manufacturing, while minimising potential risks. We have worked on various modalities, including recombinant subunit vaccines, peptides hormones, cytokines growth factors, single-domain antibodies enzymes, plasmid DNA various mRNAs, and more. We have specialized in multiple microorganisms, including Recombinant Human Collagen Production Process intracellular and extracellular secretion (yields up to 15g/L) and intracellular soluble bacteria and inclusion body (yields up to 10g/L). We have also established a BSL-2 fermentation platform to create bacteria-based vaccines. We are experts in improving processes, boosting product yields, and decreasing production costs. We have a highly efficient technology team that ensures timely and top-quality project delivery. This allows us to bring your unique products faster to market.

 EN
EN
 AR
AR
 HR
HR
 CS
CS
 DA
DA
 NL
NL
 FI
FI
 FR
FR
 DE
DE
 EL
EL
 IT
IT
 JA
JA
 KO
KO
 NO
NO
 PL
PL
 PT
PT
 RO
RO
 RU
RU
 ES
ES
 SV
SV
 IW
IW
 ID
ID
 LV
LV
 LT
LT
 SR
SR
 SK
SK
 SL
SL
 UK
UK
 VI
VI
 ET
ET
 HU
HU
 TH
TH
 TR
TR
 FA
FA
 AF
AF
 MS
MS
 BE
BE
 MK
MK
 UR
UR
 BN
BN