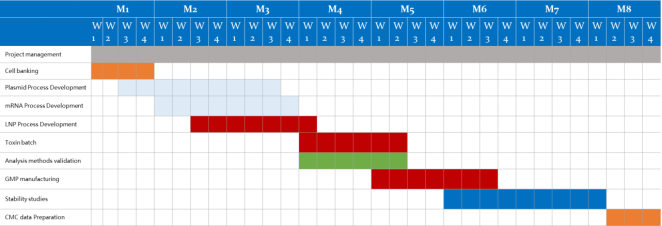
|
Process
|
Services Details
|
Unit Operation
|
|
Technology Transfer
|
Document transfer
|
Process, formulation, analytical methods and quality standard
|
|
Technical and compliance evaluation
|
Evaluation of man-machine-material-method-environment-measurement;
Evaluation of process, formulation, analytical methods and quality standard.
|
|
Technology transfer implementation
|
Manufacturing process and analytical transfer
|
|
Process validation
|
1~3 engineeringbatchesto evaluate and confirm the process is robust.
|
|
Plasmid Manufacturing
|
E. colifermentation
|
Preparation of fermentation system
|
|
Seedcultivation, fed-batch fermentation
|
|
Plasmid purification
|
E. colicell harvesting and alkaline lysis
|
|
Plasmid purification, impurities removal
|
|
Plasmid linearization
|
Single enzyme digestion
|
|
Purification of linearized template plasmid
|
|
mRNA DS Manufacturing
|
mRNA synthesis
|
In vitrotranscription (IVT) reaction
|
|
mRNA purification
|
DNA template removal
|
|
mRNA purification, impurities removal
|
|
mRNA buffer exchange
|
Tangential flow filtration
|
|
LNP DS Manufacturing
|
LNP encapsulation
|
Preparation of ethanol phase containing lipids
|
|
Microfluidics technology
|
|
Concentration and buffer exchange
|
Tangential flow filtration
|

 EN
EN
 AR
AR
 HR
HR
 CS
CS
 DA
DA
 NL
NL
 FI
FI
 FR
FR
 DE
DE
 EL
EL
 IT
IT
 JA
JA
 KO
KO
 NO
NO
 PL
PL
 PT
PT
 RO
RO
 RU
RU
 ES
ES
 SV
SV
 IW
IW
 ID
ID
 LV
LV
 LT
LT
 SR
SR
 SK
SK
 SL
SL
 UK
UK
 VI
VI
 ET
ET
 HU
HU
 TH
TH
 TR
TR
 FA
FA
 AF
AF
 MS
MS
 BE
BE
 MK
MK
 UR
UR
 BN
BN